II) Electron-Impact Excitation out of the ground state of Atoms
B. Excitation of Helium
Let us now consider the simplest multi-electron atom, helium. By the Pauli exclusion principle, the two 1s electrons in the ground state of helium have opposite spins (S=0); thus the ground state of helium is a 11S. When one of the electrons is excited to a higher level, we no longer have identical nl electrons, so both spin pairings are allowed (S=0 and S=1). Since helium is a strongly LS-coupled atom, there are no electric-dipole allowed transitions between the seperate singlet (S=0) and triplet (S=1) systems of excited states.Click here for a helium energy level diagram (GIF file: 500×600).
Excitation of singlet levels
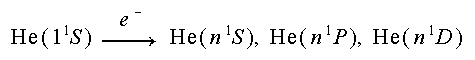
As shown below, the excitation function for excitation into the n1P levels of helium is much different than the excitation functions for the n1S and n1D levels. The n1P excitation function is broader, with a peak at a higher energy.
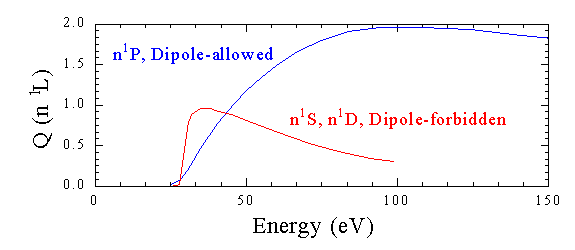
This difference can be understood by considering the radiation field produced by the incident electron as it passes the target atom. The time average of the potential will have many different multipole expansion terms. The k=1 term is analogous to a dipole-allowed transitions, the k=2 to a quadrupole transitions, etc... Thus from the known rules for optical transitions:
| k=1 | 11S -> n1P | | |
| k=2 | 11S -> n1D | | Q decreasing |
| k=3 | 11S -> n1F | |/ |
| : | ||
| k=0 | 11S -> n1S | no optical analog |
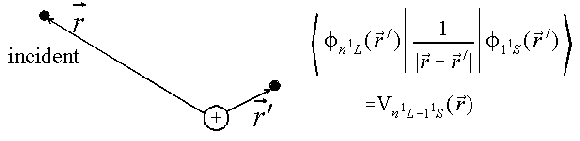 |
| The incident electron's position is r, the target atom electron that is excited is labeled by r'. The passive, non-excited electron is ignored (frozen core approximation). |
Excitation of triplet levels
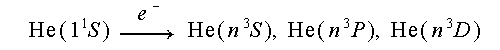
In comparison, all of the triplet excitation functions are very similar. All are sharply peaked, and decrease rapidly with increasing energy.
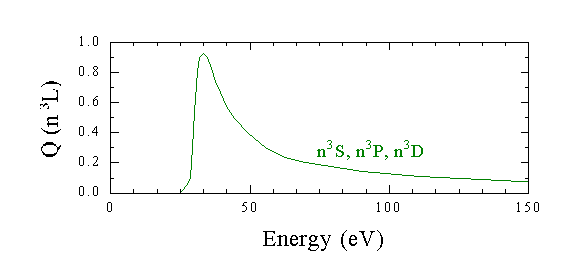
If we perform a multipole expansion, as in the case for excitation into the singlet states of helium, we now need to include the magnetic interaction terms needed to flip one of the target atom's electron spin. This process would predict cross sections much lower than those observed.
Instead, the dominant process in triplet excitation is exchange excitation: the incident electron is captured into an excited state, while one of the target atom's electrons is ejected.
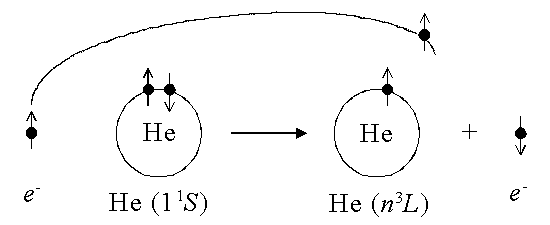
The cross section will be large only if the incident e- spends enough time around the He atom, or is moving at a low velocity. Hence the peak at the lowest possible energy, and the rapid decrease with increasing energy/velocity.
references:
Studies of Electron Excitation of Rare-Gas Atoms into and out of Metastable Levels using Optical and Laser Techniques Chun C. Lin, and L. W. Anderson, Adv. At. Mol. Opt. Phys. 29 (1991) 1-32.
 |
 |
 |