III) Excitation out of Metastable Levels of Atoms
A. Motivation
Metastable states are long lived states that are dipole-forbidden from decaying to the ground state. In comparison to ground state atoms, metastable atoms are big and fluffy (i.e. they have much larger cross sections), they also have a lot of internal energy. Since they are long lived, substantial populations of metastable atoms can form in many plasmas. Because of these properties, excitation into and out of metastable levels play a key step for many processes in plasmas, lighting discharges, atmospheric dynamics, etc...
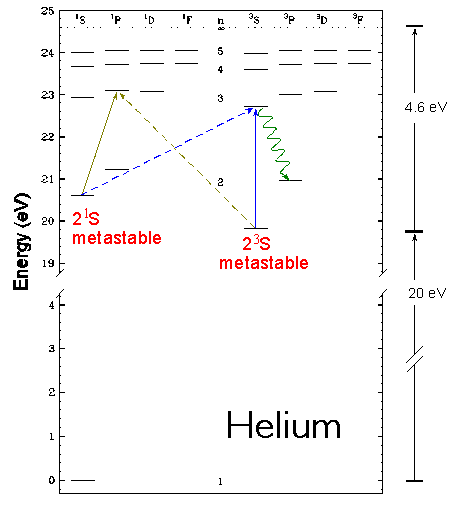
To illustrate these points, let us first examine our old friend helium. Helium has two metastable states, the 21S and the 23S. As seen in the energy level diagram, they both lie about 20eV above the ground state.
Laboratory measurements of cross sections are difficult because of the problems of producing sufficient amounts of a well characterized metastable atom target. Unlike the static gas experiments described so far, it is not possible to open a valve and fill a chamber with metastable atoms. These experiments instead must have some way of producing the metastable atoms. For the moment we will concentrate on the production of metastable helium.
Sources of Metastable Atoms
We have done experiments using two different sources of metastable atoms.- Hollow Cathode Discharge
The first experiment produces metastable atoms using a hollow cathode discharge. The discharge produces many metastable atoms, but the vast majority of atoms leaving the discharge are in the ground state. Only about 3 in 107 atoms in the target are metastable atoms. This limits the 23S-->n3L excitation measurements to energies below onset out of the ground state (i.e. less than 20eV).
The metastable target consists of a mix of both 21S and 23S atoms. In studying 23S-->n3L excitations (solid blue line in the above energy-level diagram), the 21S should contribute negilible to the signal since this is a spin-changing / exchange process (dashed blue line in diagram) and is thus small and sharply peaked.
- Charge Exchange Reaction
The second experiment uses charge exchange between helium ions and an alkali vapor to produce the metastable atoms:He+ + Cs --> He* + Cs+ This process yields a traget with more metastable atoms than ground state atoms. Thus, measurements can be taken at much higher electron energies (up to 1500eV).
Hollow Cathode Source
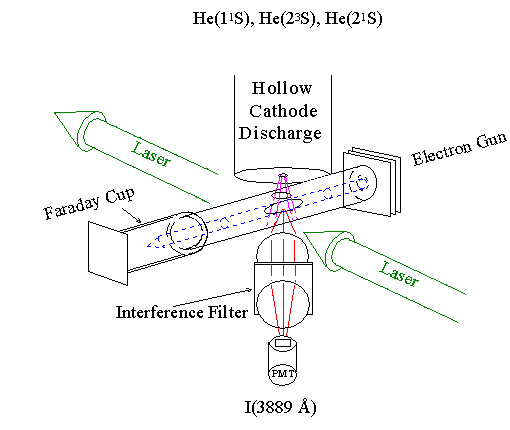
In this experimental setup a thermal beam of ground state and metastable atoms (purple) emerge from the bottom of the hollow cathode discharge. The atom beam is crossed with an electron beam (blue). The fluoresence is detected by a PMT, with wavelength selection provided by a narrow-band interference filter. To place the results on an absolute scale we use a Laser-Induced Fluoresence technique. The electron-beam excitation is replaced with excitation from a laser. The ratio of the two signals and the known optical absorbtion cross section, it is possible to determine the electron-impact excitation cross section.
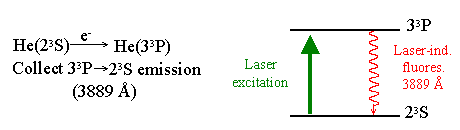
Explaination of Charge Exchange
To get around the problem of excitation out of the ground state, it would be nice to have a target composed only (or at least mostly) of metastable atoms. One way of achieving this goal is through near resonant charge exchange.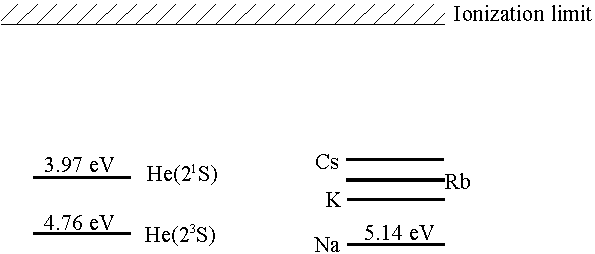
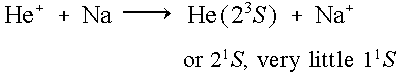
The ground state binding energy of sodium (or any of the alkali metals) is almost the same as the binding energy of the He(21S) and He(23S) levels. Thus, in a collision between a helium ion and an alkali atom, there is large cross section for charge exchange into the metastable levels (small energy defect ~0.5eV), while the cross section for charge exchange directly into the ground state is very small (large energy defect ~20eV). Nonetheless, there is some charge exchange into the He(21P) level that quickly decays to the ground state, but there are still about five metastable atoms for every one ground state atom.
| He+ + Alk --> He( 23S) + Alk+ | Energy defect ~ 0.5 eV |
| He+ + Alk --> He( 21S) + Alk+ | |
| He+ + Alk --> He( 11S) + Alk+ | Energy defect ~ 20 eV |
Thus due to the energy near-resonance, creation of both the He(23S) & He(21S) levels are favorable, while the He(11S) level is unfavorable. Using the ratio of 21S to 23S metastable atoms can be varied from 40:60 to 100:0 depending on the He+ beam energy and the alkali target used.
Fast Beam Experiment
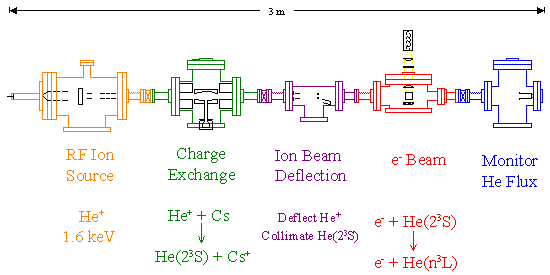
This charge exhange method is used in the fast beam experiment. A RF ion source produces a 1.6keV He+ beam. The ion beam is passed through a cesium vapor target to produce the metastable helium atoms. The remaining ions are deflected out of the way with a set of deflection plates in the next chamber, leaving only the neutral particles. The atom beam is then crossed at right angles with an electron beam, and the fluoresence is detected by a PMT at right angles to both beams. The number of atoms in the fast beam is monitored by a secondary electron detector in the last chamber.
Due to the complications of having a fast atom beam target, the absolute calibration for this experiment is a bit more complicated. Seperate experiments are preformed to determine the efficiency of the neutral detector and optical system. Information is also required on the overlap of the atom beam, the electron beam, and the optical detection region.
- Metastable Atom Beam Flux
- Metastable Atom Beam Profile
- Electron Beam Profile
- Overlap between Electron Beam & Atomic Beam
- Measure Emission Intensity from He(n3L) produced by Electron Excitation out of He(23S)
- Measure Emission Intensity from He(n3L) produced by Electron Excitation out of He(11S) ground state
references:
Studies of Electron Excitation of Rare-Gas Atoms into and out of Metastable Levels using Optical and Laser Techniques Chun C. Lin, and L. W. Anderson, Adv. At. Mol. Opt. Phys. 29 (1991) 1-32.
Cross sections for electron excitation of the 23S metastable level of He into higher triplet levels Garrett A. Piech, Mark E. Lagus, L. W. Anderson, Chun C. Lin, and M. R. Flannery, Physical Review A 55 (1997) 2842-2856.
A method for measuring cross sections for electron-impact excitation out of metastable levels of atoms Ronald B. Lockwood, L. W. Anderson, and Chun C. Lin, Z. Phys. D- Atoms, Molecules and Clusters 24 (1992) 155-160.
Apparatus for measuring electron-impact excitation cross sections using fast metastable atoms produced via charge exchange John B. Boffard, Mark E. Lagus, L. W. Anderson, and Chun C. Lin, Rev. Sci. Instrum. 67 (1996) 2738-2751.
 |
 |
 |